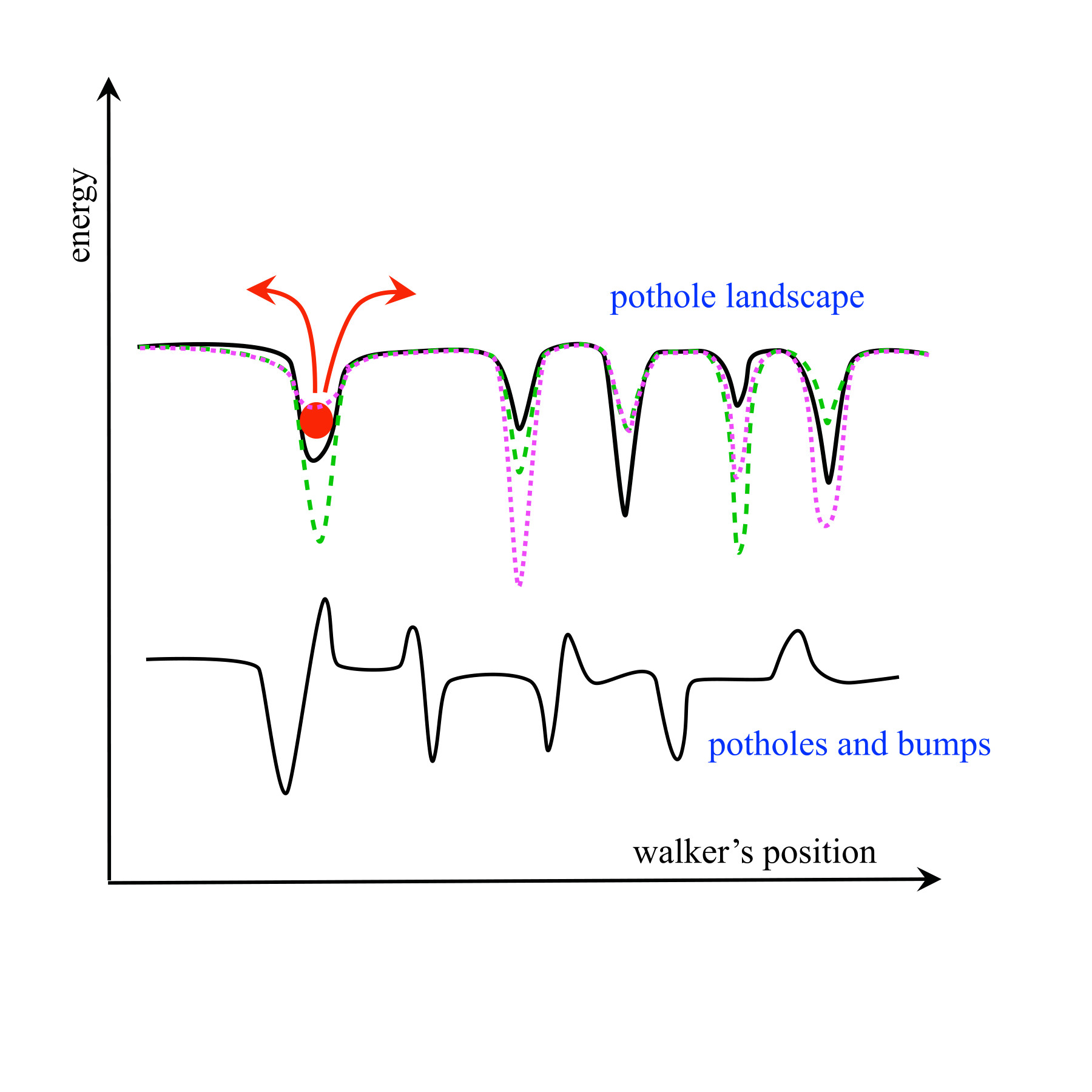
Inside of a living cell, there is no such thing as moving through empty space. Molecules inside drift and collide, constantly feeling forces from everything around them, such as electric charges or nearby structures. In a basic liquid like water, scientists can describe the motion of such molecules surprisingly well using the theory of Brownian motion, which maintains that every microscopic detail about the liquid isn’t required for accurate predictions. Instead, only a few key properties are needed, such as temperature and viscosity. While this theory does work, a cell is more complex and unpredictable than water. So, this raises the question: can existing frameworks still be applied, or does a new framework need to be developed to explain how molecules move in environments like these?
This question sits at the heart of a new study by Dmitrii E. Makarov, core faculty member at the Oden Institute for Computational Engineering and Sciences, and a professor in the Department of Chemistry at The University of Texas at Austin and his co-author Peter Sollich, a professor of theoretical physics at the University of Göttingen in Germany.
Their paper, “Static and Dynamic Rough Energy Landscapes Can Lead to Identical Diffusivity,” published in the Proceedings of the National Academy of Sciences, explores a counterintuitive idea: even if the “obstacles” slowing molecular movement in living cells fluctuate rapidly, motion may not get faster. In some cases, it actually slows down exactly as much as if nothing were fluctuating at all.